Professor Qingzhong Xiao
/filters:format(webp)/prod01/channel_144/whri/media/the-william-harvey-research-institute/staff-and-students/research-staff/Xiao,-Q.jpg)
Chair of Stem Cell and Cardiovascular Disease
Centre: Clinical Pharmacology and Precision Medicine
Email: q.xiao@qmul.ac.ukTelephone: +44(0) 20 7882 6584
Profile
ORCID iD: 0000-0001-9101-0498
I obtained my Bachelor’s degree in medicine from Chongqing University of Medical Sciences, China, in 1994. Subsequently, I worked as clinical teaching fellow at the Department of Laboratory Medicine, Guangzhou Medical College and was promoted to Lecturer in Clinical Microbiology and Immunology in 1999. In the meantime, I obtained my Master’s Degree in Medical Immunology in 1999 and my Doctoral Degree (PhD) in Pathology and Pathophysiology in 2003 at Sun Yat-Sen University, Guangzhou, China. I joined Professor Qingbo Xu’s group at St Georges’ between 2003 and 2006 before moving to King’s College London (2006-2009), University of London, UK where I worked as a postdoctoral research fellow and research associate. I established novel approaches to successfully induce embryonic stem cell differentiation towards vascular endothelial cells and smooth muscle cells. I successfully applied these techniques to investigate the therapeutic effects of stem cell-derived vascular cells in cardiovascular diseases such as damaged/injured vessels.
In 2010, as the recipient of British Heart Foundation (BHF) Intermediate Basic Science Research Fellowship I joined William Harvey Research Institute (WHRI) to expand my research ambition in stem cell biology, genetics and cardiovascular diseases. With the support of my BHF fellowship and project grants, I was able to set up my independent research laboratory (Stem Cell and Cardiovascular Disease) at WHRI. I was promoted to Senior Lecturer and Reader in Stem Cell and Cardiovascular Disease in 2013 and 2016, respectively before becoming Professor (Chair) of Stem Cell and Cardiovascular Disease in 2018.
My long-term research aim is to uncover critical signal pathways that govern vascular development, identify key molecules that control cardiovascular disease progression and develop them into a more effective therapeutic options for cardiovascular disease.
Research
Current Group Members:
Xu Wang (PhD, PDRA), Meixia (MD, PhD, Visiting Associate Professor), Haotian Zhao (PhD candidate), Leyu Wang (PhD Associate), Xiaoyue Zhou (PhD Associate), Xiangyu Wang (PhD candidate), Stephane Bourgeois (PhD, Lab Manager), Mrs. Denise Grant (Personal Assistant and Centre Administrator)
Previous PDRAs:
Dr Elisa Ferraro, (PDRA, 2024-2025), Dr Weiping Lin (PDRA, 2024), Dr Caroline Silveira Martinez (PDRA, 2021-2024), Dr Wei Wu (PDRA, 2018-2019), Dr Weiwei An (PDRA, 2015-2019), Dr Iliana Fauzi (PDRA, 2016-2018), Dr Le Anh Luong (PDRA, 2013-2017), Dr Guanmei Wen (PDRA, 2012-2015)
PhD Students:
Dr Chenxin Liu (PhD student; Awarded: 2025), Dr Kaiyuan Niu (PhD student; Awarded: 2024), Dr Eithne Margaret Maguire (PhD student; Awarded: 2021), Dr Shiping He (PhD student; Awarded: 2020), Dr Stuart William Alfred Pearce (PhD student; Awarded: 2020), Dr Tayyab A. Afzal (PhD student; Awarded: 2019), Dr Xiaotian Yu (PhD student; Awarded: 2015), Dr Hanqing Zhao (PhD student; Awarded: 2015), Dr Gang Wang (PhD student; Awarded: 2011), Dr Zhenling Luo (PhD student; Awarded: 2011), Dr Anna E. Pepe (PhD student, Awarded: 1999)
PhD Student Associates:
Dr Ancheng Zheng (PhD Associate, Awarded: 2025), Dr Zhisheng Yang (PhD Associate; Awarded: 2024), Dr Xinmiao Zhou (PhD Associate; Awarded: 2022), Dr Mei Yang (PhD Associate; Awarded: 2020), Dr Feng Yang (PhD Associate; Awarded: 2017), Dr Qishan Chen (PhD Associate; Awarded: 2015), Dr Yuan Huang (PhD Associate; Awarded: 2014)
Clinical and Academic Visitors:
Dr Shiyuan Huang (Academic Visitor, 2023-2024), Dr Chongkai Fang (Academic Visitor, 2022-2023), Dr Silin Liu (Academic Visitor, 2022-2023), Dr Xiaolei Sun (Academic Visitor, 2020), Dr Jun Luo (Academic Visitor, 2019-2020), Dr Renqiang Yang (Academic Visitor, 2019), Dr Jiangyong Chen (Academic Visitor, 2016-2017), Dr Dan Chen (Academic Visitor, 2015-2016), Dr Cheng Zhang (Academic Visitor, 2014~2015, 2020), Dr Luyang Lin (Academic Visitor, 2012-2013)
Summary
- Vascular stem cell biology and differentiation
- Pluripotent stem cells (ESCs, embryonic stem cells; iPSCs, induced pluripotent stem cells)
- Vascular biology and diseases (Atherosclerosis; Post-angioplasty restenosis or In-stent restenosis; Aortic aneurysm and dissection; Vascular calcification; Transplant atherosclerosis; etc.)
- Cardiac diseases (Heart failure with preserved ejection fraction; Myocardial infarction; Myocardial ischemia-reperfusion (I/R) injury; etc.)
- Cardiovascular regenerative medicine
- Stem cell-derived vascular and cardiac organoids (human iPSCs)
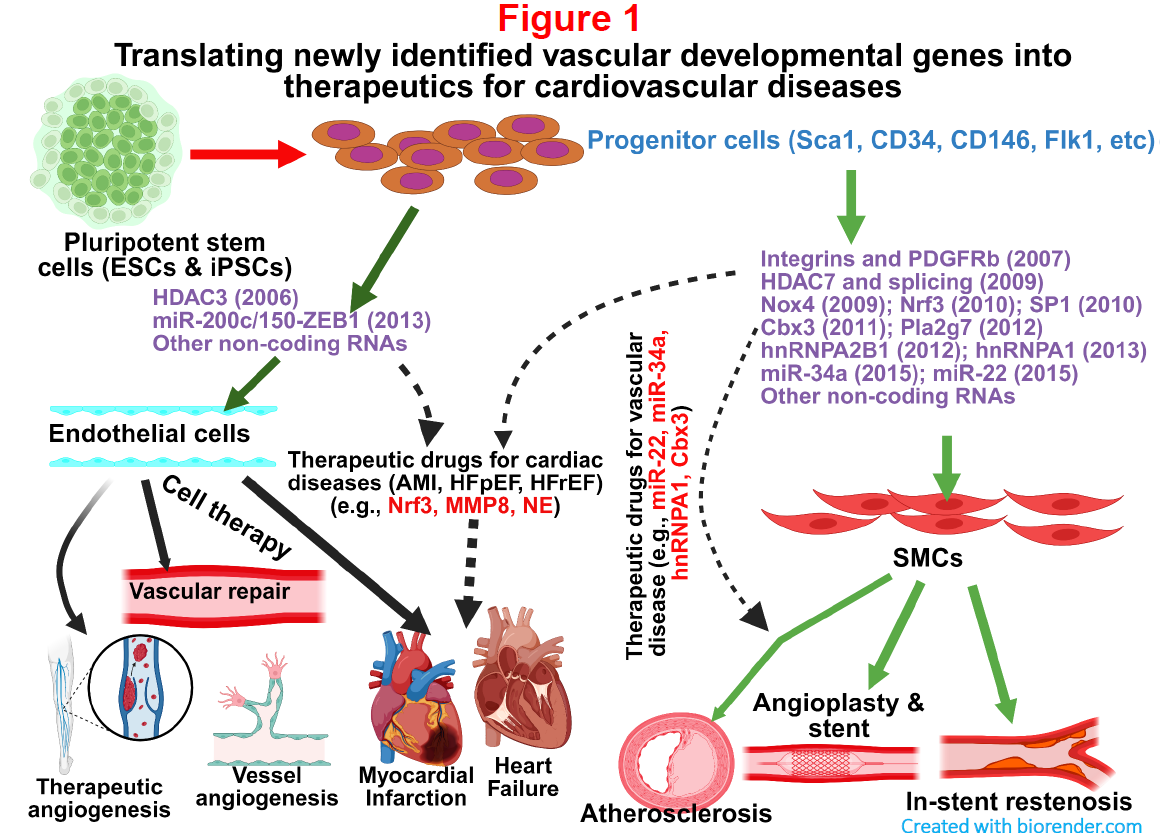 1. Unravelling novel molecular mechanisms underlying cardiovascular cell differentiation from stem/progenitor cells, and exploring their functional implications and therapeutic potential in cardiovascular diseases (CVD).
1. Unravelling novel molecular mechanisms underlying cardiovascular cell differentiation from stem/progenitor cells, and exploring their functional implications and therapeutic potential in cardiovascular diseases (CVD).
One of my main research interest is specifically focused on the study of identifying novel targets and signalling pathways such as microRNAs, transcription factors and other molecules which are crucial for vascular endothelial cell (EC) and smooth muscle cell (SMC) differentiation from murine/human pluripotent stem cells as well as adult stem/progenitor cells. By establishing simple but efficient vascular cell differentiation models, I was the first to report that matrix protein Collagen-IV, Nox4, Nrf3, Pla2g7, HDAC3, HDAC7, Cbx3, hnRNPA1, hnRNPA2B1, microRNA-34a, and miR-22 play a key regulatory role in vascular cell differentiation and vascular development (Figure 1). My goal is to continue exploring the key signal pathways underlying cardiovascular cell differentiation and cardiovascular development and their implications in cardiovascular diseases. Such findings will provide useful mechanistic insights into stem cell differentiation towards cardiovascular cells and will significantly enhance the knowledge of stem cell biology and cardiovascular biology in key areas of stem cell self-renewal, vasculogenesis, angiogenesis, and cardiovascular repair.
2. Uncovering the contributions of stem/progenitor cells (SPCs) to cardiovascular diseases (CVD).
Atherosclerosis is the underlying cause of CVDs with growing evidence indicating that SPCs play a pivotal role in the development of atherosclerosis and other CVDs. My group’s ongoing aims is to study the contribution of multiple SPCs to the pathogenesis of CVDs. Using the combination of animal models, human tissue samples, single cell RNA sequencing (scRNA-seq) analysis and cellular lineage in vivo tracing, we aim to uncover the novel mechanisms underlying SPC cell fate decision in the context of CVD pathogenesis and investigate potential uses of stem cell therapy for vascular disease.
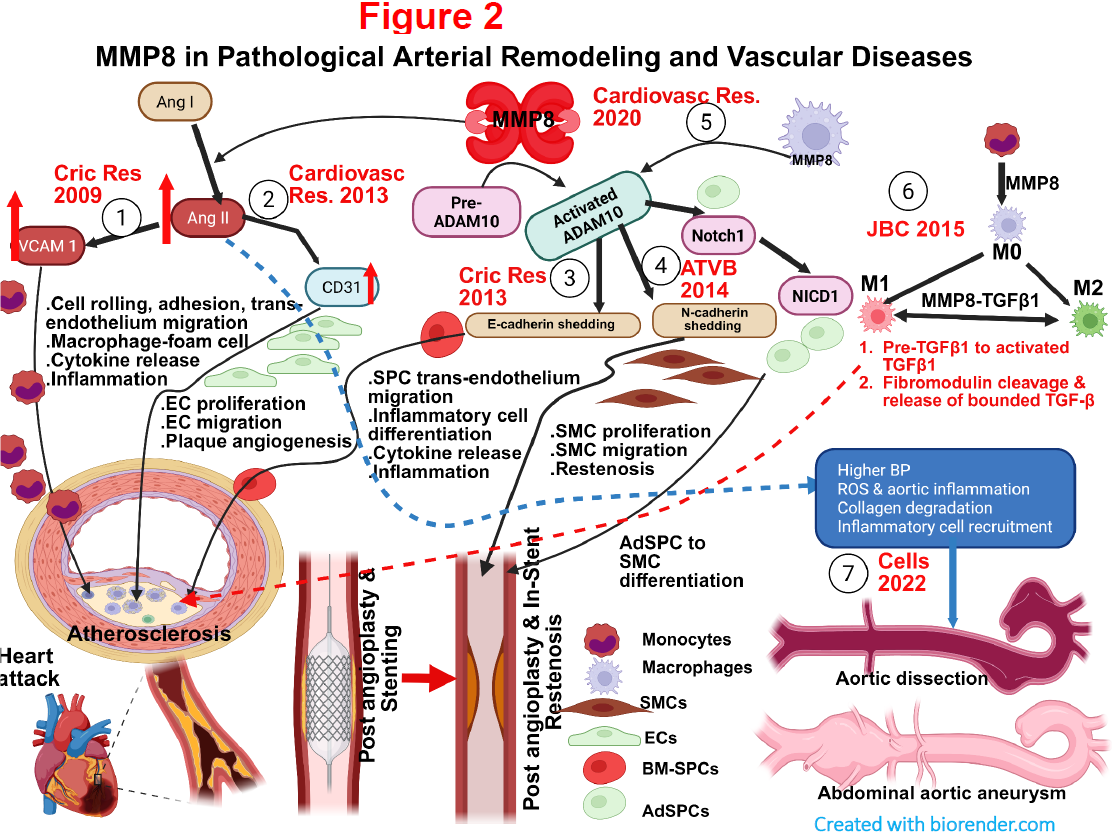 3. Identifying and exploring the potential novel therapeutic targets for cardiovascular diseases.
3. Identifying and exploring the potential novel therapeutic targets for cardiovascular diseases.
Despite major advances over the past decade, CVD is still the most common cause of death worldwide. Accumulating evidence indicates that different proteases such as matrix metalloproteinases (MMPs) and neutrophil elastase (NE) play an important and distinct role in the development of atherosclerotic lesions and atheromatous plaque rupture. However, their functional role and underlying molecular mechanism remains incompletely understood. One of the key focus of my group is the investigation into the therapeutic potential of MMP8 and NE in various CVDs and elucidating its associated molecular mechanisms (Figure 2). Moreover, by using animal models of CVDs, we were able to confirm dysregulation of key vascular development genes/molecules (e.g. miR-34a, miR-22, Nrf3, and hnRNPA1) identified from our aforementioned stem cell differentiation studies represent potential novel therapeutic candidates for CVDs. Moving forward, we aim to utilise similar strategies to continue identifying and exploring additional novel therapeutic targets for CVDs.
4. Functional involvements of Non-coding RNAs in stem cell fate decision and vascular diseases.
Recently, growing evidence has suggested that non-coding RNAs including microRNAs, long non-coding RNAs, and circular RNAs play crucial roles in embryonic development and various diseases. I aim to explore the significance of these molecules in stem cell pluripotency and vascular cell specifications in addition to their application in preventing vascular diseases.
5. Cellular reprogramming and cardiovascular regeneration.
Cellular reprogramming of somatic cells into pluripotent stem cells or other somatic cells has opened the new avenues of biomedical research and regenerative medicine. Endothelium dysfunction or damage is a hallmark of the onset of vascular diseases. Cell therapy strategies that aim to rapidly repair and restore vascular function are being increasingly explored as viable therapeutic avenues. Development of efficient and robust new methodologies that produce well-characterised, homogenous, clinical-grade cells suitable for tissue repair/re-modelling will have great utility. By using molecules discovered by my group’s early stem cell studies I aim to investigate their potential utility in reprogramming somatic cells into functional vascular cells with clear implications in cardiovascular regeneration and treatment.
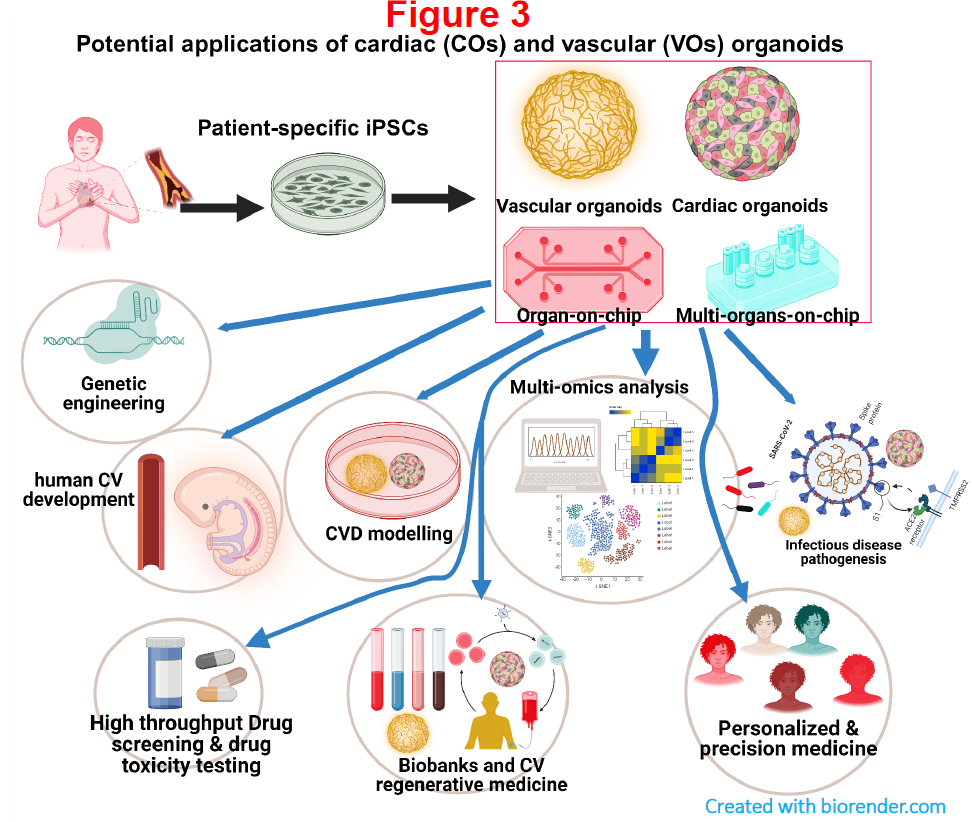 6. Establishing novel cardiac and vascular organoids as well as heart/vessel-on-chip from human iPSCs for personalized cardiovascular medicine.
6. Establishing novel cardiac and vascular organoids as well as heart/vessel-on-chip from human iPSCs for personalized cardiovascular medicine.
Traditional models including 2D cellular models and animal models have intrinsic and inherent limitations in fully capturing the intricacies of human tissue pathophysiology thereby reducing translational value and drug development potential. Recent advances has led to the development of cardiac and vascular organoids and heart/vessel-on-chip platforms. These novel stem cell based platforms can closely mimic the human cardiovascular system and pathophysiology, giving rise to high fidelity models that are superior to conventional models. Accordingly, my group seeks to develop novel cardiac and vascular organoids alongside heart/vessel-on-chip platforms from human iPSCs. Together with patient-specific iPSCs and gene editing tools, we hope to uncover disease aetiologies, investigate therapeutic interventions and conduct drug screening in a physiologically relevant context for the eventual goal of offering a personalized treatment for CVD (Figure 3).
Publications
-
Xiao Q, Yang M, Zhang L et al. (2026). Neutrophil Elastase: A Novel Therapeutic Target for Heart Failure with Preserved Ejection Fraction. nameOfConference
-
Mou W, Shan W, Yu S et al. (2025). Interpretable machine learning model for predicting recurrence in patients with diabetic foot ulcers. nameOfConference
-
Zheng A, Yang M, Martinez C et al. (2025). Abstract 4357158: Neutrophil Elastase: A Novel Therapeutic Target for Heart Failure with Preserved Ejection Fraction. nameOfConference
QMRO: qmroHref -
Xiao R, Darr H, Khan Z et al. (publicationYear). Updated Applications of Stem Cells in Hypoplastic Left Heart Syndrome. nameOfConference
QMRO: qmroHref -
Zheng A, Yang M, Silveira Martinez C et al. (2025). BS53 Neutrophil elastase: a novel therapeutic target for heart failure with preserved ejection fraction. Basic science
QMRO: qmroHref -
Sun X, Wu J, He T et al. (2025). Mitochondrial One-Carbon Metabolism Drives CD34-Lineage Cells to Differentiate Into T Follicular Helper Cells to Form Tertiary Lymphoid Organs in Transplant Arteriosclerosis. nameOfConference
-
Chen Q, Sun S, Shi Z et al. (2025). Novel roles of Nrf3–Trim5 axis in vascular smooth muscle cell dysfunctions and neointimal hyperplasia. nameOfConference
DOI: 10.1093/cvr/cvaf084
QMRO: qmroHref -
Chen Q, Zheng A, Xu X et al. (2025). Nrf3-Mediated Mitochondrial Superoxide Promotes Cardiomyocyte Apoptosis and Impairs Cardiac Functions by Suppressing Pitx2. nameOfConference
-
Tang Y, Wu J, Sun X et al. (2025). Cardiolipin oxidized by ROS from complex II acts as a target of gasdermin D to drive mitochondrial pore and heart dysfunction in endotoxemia. nameOfConference
QMRO: qmroHref -
An W, Luo J, Zhang C et al. (2025). Integrative Bioinformatics Analysis to Identify Key Ferroptosis-Related Genes and Immune Infiltration in Aortic Aneurysm and Dissection: Implication of PTGS2. nameOfConference
DOI: 10.2147/jir.s488651
Sponsors
Collaborators
Internal
- Professor Qingbo Xu (WHRI)
- Professor Andrew Tinker (WHRI)
- Professor Paul Charles Evans (WHRI)
- Professor Thomas Iskratsch (SEMS)
External
- Professor Li Zhang, Xinhua Hospital affiliated to Shanghai Jiao Tong University School of Medicine, Shanghai, China
- Professor Ye Shu, University of Leicester, UK and Yong Loo Lin School of Medicine, National University of Singapore, Singapore
- Professor Chengxin Zhang, First Affiliated Hospital of Anhui Medical University, Anhui, China
- Professor Yi Yan, The Third Affiliated Hospital, Guangzhou Medical University, Guangzhou, China
- Professor Jingjing Cai, the Third Xiangya Hospital, Central South University, Changsha, China
- Professor Qingchen Wu, The First Affiliated Hospital of Chongqing Medical University, Chongqing, China
Teaching
Undergraduate Education:
- BSc Pharmacology and Innovative Therapeutics (BScPhIT): Lab-based research project supervisor; BSc Pharmacology - Project Skills in the Life Sciences (BIO603) supervisor; BSc Biomedical Science - Biomedical Science Research Project (BMD600) project supervisor
- MBBS: SSC supervisor; Academic supervisor; PBL facilitator
Postgraduate Education:
- MSc in Regenerative Medicine: Lecturer; Project supervisor
- BHF MRes: Lecturer and Journal club
- PhD supervisor and examiner
External Education Activities:
- PhD Examiner: UK and China